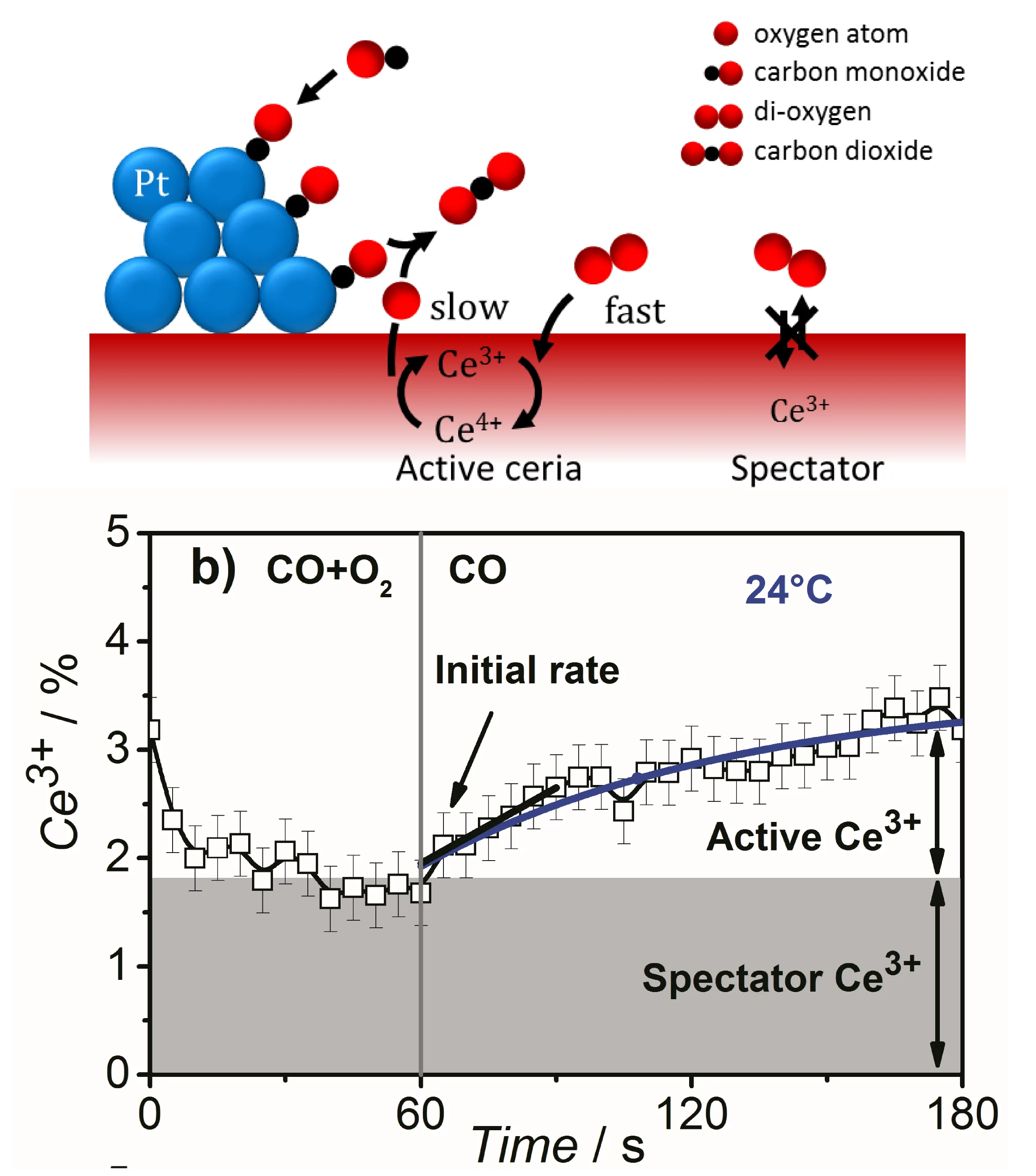
Identification of active species and the rate-determining reaction steps are crucial for optimizing the performance of oxygen-storage materials, which play an important role in catalysts lowering automotive emissions, as electrode materials for fuel cells, and as antioxidants in biomedicine. We demonstrated that active Ce3+ species in a ceria-supported platinum catalyst during CO oxidation are short-lived and therefore cannot be observed under steady-state conditions. Using time-resolved resonant X-ray emission spectroscopy, we quantitatively correlated the initial rate of Ce3+ formation under transient conditions to the overall rate of CO oxidation under steady-state conditions and showed that ceria reduction is a kinetically relevant step in CO oxidation, whereas a fraction of Ce3+ was present as spectators. This approach can be applied to various catalytic processes involving oxygen-storage materials and reducible oxides to distinguish between redox and nonredox catalytic mechanisms.
Contact
Dr Olga SafonovaSuperXAS beamline
Laboratory for Synchrotron Radiation and Femtochemistry (LSF)
Swiss Light Source, Paul Scherrer Intitute
5232 Villigen-PSI, Switzerland Telephone: +41 56 310 58 05
E-mail: olga.safonova@psi.ch
Original Publication
Catalytically Active and Spectator Ce3+ in Ceria-Supported Metal CatalystsRene Kopelent, Jeroen A. van Bokhoven, Jakub Szlachetko, Jacinta Edebeli, Cristina Paun, Maarten Nachtegaal, and Olga V. Safonova
Angewandte Chemie, 9 June 2015
DOI: 10.1002/ange.201503022