In an international collaboration between the University of the British Columbia (UBC) and the Paul Scherrer Institut (PSI), a promising system for painless and minimally-invasive therapeutic drug monitoring has been demonstrated. Results of the study have been published in the latest issue of the nature-journal “scientific reports”.
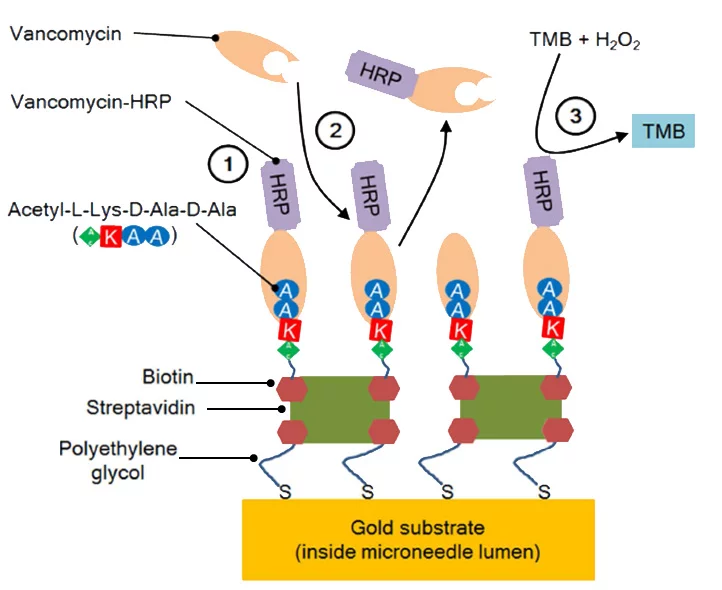
The proposed device is based on the combination of an optofluidic system with hollow microneedles – which were developed and fabricated at UBC – to extract extremely small volumes (< 1 nL) of interstitial fluid (ISF) to measure drug concentrations. The inner lumen of the microneedles was first functionalized for the detection of vancomycin using an enzyme-linked displacement assay. Quantification was achieved via absorbance measurements through optical fibres coupled to the optofluidic device.
The enzyme assay combined with the optofluidic sensing system for vancomycin detection provides high sensitivity clinically relevant ranges, a low limit of detection (< 100 nM) determined in extremely low sample volumes (0.6 nL), and rapid measurements (< 5 min in total) of drug binding to microneedles.
Using vancomycin as a target drug, the potential of the microneedle-optofluidic biosensor for therapeutic drug monitoring (TDM) was demonstrated for point of care applications using a minimally invasive sample extraction and limited needs for external equipment (only a diode laser and a photodetector were required). These results prove the potential of the proposed integrated sensors for the development of portable devices that can be used by patients to perform TDM with many drug candidates present in ISF.
Contact
Dr. Victor CadarsoLab. for Micro- and Nanoechnolgy
Paul Scherrer Institut
Telephone: +41 56 310 5146
E-mail: victor.cadarso@psi.ch
Original Publication
Integrated hollow microneedle-optofluidic biosensor for therapeutic drug monitoring in sub-nanoliter volumesSahan A. Ranamukhaarachchi, Celestino Padeste, Matthias Dübner, Urs O. Häfeli, Boris Stoeber, Victor J. Cadarso
Scientific reports 2016, 6, 29075; DOI: 10.1038/srep29075
Media Releases
UBC Media Release | July 25, 2016EurekAlert Public Release | July 25, 2016